|
Home
Definition
Treatment
Genetics
The Gunn rat
Bilirubin toxicity
Phototherapy
Crigler-Najjar and pregnancy
Crigler-Najjar type II
Bilirubin Levels & Drugs
International Crigler-Najjar Registry and Associations
Links
News
Album
Updates
|
|
 PREGNANCY in CRIGLER-NAJJAR SYNDROME:
PREGNANCY in CRIGLER-NAJJAR SYNDROME:
The exact prevalence of Crigler-Najjar disease is unknown. It is estimated to be below 1 case for 1 million live births. In some populations (for example in Amish and Mennonites) prevalence is higher.
Mutated gene frequency for autosomal recessive disorders is estimated by calculating the square root of the disease frequency. Considering that Crigler-Najjar disease occurs in 1 in 1,000000, the altered gene frequency is 1:1000. Therefore, the frequency of unaffected persons carrying the mutated UGT1A1 gene (defined as carrier or heterozygote) is 1:500, since we have two copies of each autosomal gene.
Carriers for a mutation in the Crigler-Najjar syndrome gene have about half the UGT1A1 enzyme activity of a normal adult. Despite that, since along with the mutated copy they have a copy of the "correct" gene, they do not have significant consequences in the bilirubin levels.
With genetic testing (see below) it is possible to determine whether a person has a mutation in the UGT1A1 gene.
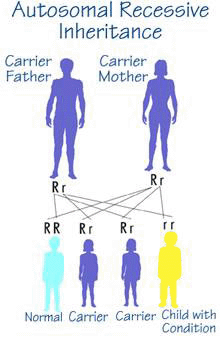
A Crigler-Najjar patient (homozygote) has two parents who are unaffected but each parent carries an altered (mutated) copy of the UGT1A1 gene along with a "correct" copy of the gene (heterozygote).
The risk for two heterozygotes (carriers) to have an affected child is 25%, 1 in 4, for each child that they have. In this case the child takes the mutated copy of the gene from each parent. Similarly, there is a 3 in 4 chance (75%) that each child will not be affected by the disease.
Males and females are at equal risk for being affected by Crigler-Najjar syndrome.
Before the use of phototherapy as therapeutic option, Crigler-Najjar syndrome type I was fatal during childhood. Nowadays, patients compliant with this kind of treatment can reach adulthood.
Whether followed by constant medical monitoring Crigler-Najjar patients may successfully give birth to healthy babies.
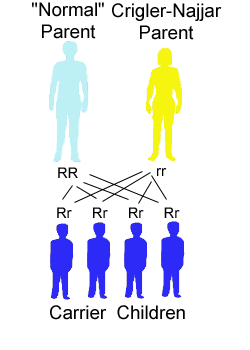
The risk for a Crigler-Najjar patient (homozygote) having a baby with a unaffected (not carrier) person to have a child affected with the disease is 0%. All children will be carrier (heterozygote) with a copy of the mutated gene and a copy of the correct gene.
As said above, in general population (i.e. in absence of a family history of Crigler-Najjar syndrome) the chance for an unaffected person to have a mutation in the UGT1A1 gene (in other words to be a carrier of the disease) is 1:500.
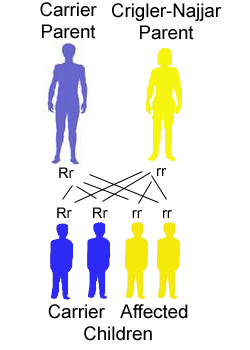
The risk for a Crigler-Najjar patient (homozygote) having a baby with an unaffected carrier (heterozygote) person to have a child affected with the disease is 50%, for each child they have. There is a remaining 50% chance that the children will be unaffected carriers.
It has been reported that hyperbilirubinemic female Gunn rats
(the animal model of Crigler-Najjar syndrome) have reduced fertility (Davis D.R., Yeary R.A. Impaired fertility in the jaundiced female Gunn rat. Lab Anim Sci. 1979;29:739-43). No data are available on fertility of Crigler-Najjar patients.
Here we give links to several clinical reports dealing with patients affected by Crigler-Najjar syndrome during pregnancy.
Case Reports:
-
Chaubal et al. Management of pregnancy in Crigler Najjar syndrome type 2. World J Hepatol 2016; 8(11): 530-532
Abstract
Full Text
-
Chaudhary et al. A rare case of Crigler-Najjar syndrome type II with pregnancy. Int J of Reprod Contracept Obstet Gynecol 2014; 3(1): 261-262
Abstract
Abstract
Full Text
-
Passuello et al. Pregnancy outcome in maternal Crigler-Najjar Syndrome Type II: a case report and systematic review of the literature.
Fetal Diagnosis and Therapy, 2009.
Full Text
-
Arora N, Choudhary S. Pregnancy with Crigler-Najjar syndrome type II. Journal of Obstetrics and Gynaecology, 2009; 29: 242-244.
Abstract
-
Arora N, Choudhary S. Pregnancy with Crigler-Najjar syndrome type II. Journal of Obstetrics and Gynaecology, 2009; 29: 242-244.
Abstract
-
Hannam S, Moriaty P, O’Reilly H, Craig JS, Heneghan MA, Baker A and Dhawan A. Normal neurological outcome in two infants treated with exchange transfusions born to mothers with Crigler-Najjar Type 1 disorder. European Journal of Pediatrics. 2008; 16: 427-429.
Abstract
Full Text
-
Gajdos V, Petit F, Trioche P, Mollet-Boudjemline A, Chauveaud A, Myara A, Trivin F, Francoual J, Labrune P. Successful pregnancy in a Crigler-Najjar type I patient treated by phototherapy and semimonthly albumin infusions. Gastroenterology. 2006; 131: 921-924.
Full Text
-
Pinkee S, Renu A, Bharati M. Crigler Najjar syndrome with pregnancy. J. Obstet. Gynaecol. India. 2005; 55: 270-271.
Full Text
-
Holstein A, Plaschke A, Lohse P, Egberts EH. Successful photo-and phenobarbital therapy during pregnancy in a woman with Crigler-Najjar syndrome type II. Scand J Gastroenterol. 2005; 40: 1124-1126.
Abstract
Full Text
-
Ito T, Katagiri C, Ikeno S, Takahashi H, Nagata N, Terakawa N. Phenobarbital following phototherapy for Crigler-Najjar syndrome type II with good fetal outcome: a case report. J Obstet Gynaecol Res. 2001; 27: 33-35.
Abstract
-
Smith JF, Jr., Baker JM. Crigler-Najjar disease in pregnancy. Obstet Gynecol. 1994; 84: 670-672.
Abstract
-
Taylor WG, Walkinshaw SA, Farquharson RG, Fisken RA, Gilmore IT. Pregnancy in Crigler-Najjar syndrome. Case report. Br J Obstet Gynaecol. 1991; 98: 1290-1291.
Abstract
Please, remember that these informations are not intended to be a substitute for professional medical advice.
Seek genetic counseling if you are planning a pregnancy and you are affected or have a family history of Crigler-Najjar syndrome.
Molecular diagnosis for Crigler-Najjar syndrome:
Genetic testing, including prenatal diagnosis and preimplantation genetic diagnosis , for Crigler-Najjar syndrome type I and type II patients and relatives are possible.
To find laboratories specialized in molecular diagnosis refer to the following websites:
- Orpha.net database
- US National Center for Biotechnology Information
- European Directory of DNA diagnostic Laboratories
- EuroGentest
- Gentests
- Sir Ganga Ram Hospital, New Delhi, India.
- Clinic for Special Children, Strasburg, PA.
- Mayo Clinic, Rochester, MN.
- Genesis Genetics (preimplatation genetic diagnosis).
- Australian and New Zealand Laboratories.
- Eastern Biotech. Genetic testing in Dubai and Middle East
 
PREVIOUS PAGE NEXT PAGE

|




